Embryonic stem cells (ES cells) are remarkable. They come from animal (including human) embryos and can morph into any cell in the body such as brain, bone marrow, intestine, muscle, or blood cells. Biologists call them pluripotent and can isolate them from an embryo and grow them in laboratory petri dishes. In the halcyon days of early stem-cell research, it did not escape the attention of scientists that genetic changes could be made to an ES cell, the cell could then be inserted back into an embryo, and the embryo placed into the womb where it would differentiate into all the cells of the body with the new genetic modification. The process became so widespread in the early 1990s that biologists referred to the genetically modified animals born as transgenic. An example that caught the attention of the world was a mouse that had a gene from a jellyfish that made it glow in the dark (under blue lamps). It was as if a grand gift had been given to geneticists that enabled them to understand how genes functioned. Mice could be made to double in size, develop Alzheimer’s disease, grow cancer tumors, age prematurely, increase memory, or erupt with epilepsy all through gene manipulation. It was a remarkable way for scientists to study genetic diseases. There was just one problem — human ES cells did not respond favorably to genetic modifications the way mouse ES cells did. There would be no transgenic humans anytime soon even if ethical issues were overcome.
 Embryonic mouse stem cells
Embryonic mouse stem cells
Meanwhile, geneticists were probing a myriad of other ways to correct specific genetic disorders. One group focused on a gene called ornithine transcarbamylase (OTC) which codes for an enzyme that breaks down proteins in the liver. Without the enzyme, a product of the protein, ammonia, accumulates throughout the body. As you might imagine, ammonia buildup in the body can have devastating consequences, and most children do not survive into adulthood with the genetic disorder. Enter Jesse Gelsinger who had a mild case of OTC deficiency1. Mark Batshaw and James Wilson, then at the University of Pennsylvania, postulated that they could add the OTC gene to a cell’s DNA from Gelsinger and insert it back into his body via an adenovirus (viruses reproduce by entering the body and injecting either DNA or RNA into a living cell, effectively taking over the cell to reproduce more copies of themselves). The hope was that the virus would insert the corrected DNA into Gelsinger’s liver cells which would then synthesize the requisite enzyme needed by Jesse. The treatment worked in mice but had mixed results in monkey trials — some monkey immune systems responded in drastic ways causing liver failure and other disorders. Batshaw and Wilson responded by making the virus less potent and reducing the dose for the proposed trial with Gelsinger. In 1997, they approached the Recombinant DNA Advisory Committee (RAC) of the government’s National Institute of Health for approval. RAC agreed, and Jesse and his father were excited volunteers, convinced that Jesse’s close encounters with death from food reactions, regimented diet, and the plethora of pills he took was coming to an end.
On September 9, 1993, Jesse began his trial of viral injections. Four days later he was dead from a massive immune reaction to the virus. The press reports set off a chain reaction energizing Congress to initiate hearings, district attorneys investigating, the university back pedaling, and official inquiries launched by the FDA and RAC. When it was discovered that there was a “pattern of neglect” with the research by Batshaw and Wilson, the FDA halted all trials in other laboratories and a strict moratorium fell over the entire research discipline. We will never know how much of the response was an effort to point blame away from governmental agencies, but Batshaw and Wilson became the “fall guys” and genetic research would be impacted for a decade. I recognize the need for caution and ethical considerations, but I also know that there are children dying from diseases like OTC every day. I am sure many of them and their parents would gladly accept the chance of survival beyond childhood via potentially risky experiments. Did we throw the baby out with the bath water? After all, Jesse died wanting to help others by finding a cure for OTC. He did not die from the trial’s basic premise. He died because his body had highly reactive antibodies to the virus because he had probably been exposed to a similar adenovirus in his past.
Fortunately, Jesse’s disturbing death did not affect genetic diagnosis – attributing genes to diseases. Examples include the BRCA1 gene associated with breast cancer, CNV mutations linked to schizophrenia, and ADCY5 and DOCK3 genes related to neuromuscular disease. I highly recommend Siddhartha Mukherjee’s new book entitled The Gene: An Intimate History2 for further reading.
But to set the stage for the technology available today, we need to look at in vitro fertilization (IVF). In IVF, an embryo is formed from the fertilization of an egg by a sperm outside of the body. The single-cell embryo is bathed with nutrient-rich fluids in an incubator and left to divide for three days until there are 8 to 16 cells. The embryo is then implanted into a woman’s womb. Remarkably, if a few cells are removed from the growing embryo in the incubator, the embryo is unaffected. It simply replaces the lost cells. Usually several eggs are harvested for IVF and fertilized. Cells can then be removed from each embryo and genetically tested or screened for mutations allowing only a fertilized egg with no known serious genetic disorders to be implanted in the womb. Genetic testing in this way has been done since the late 1980s and is referred to as preimplementation genetic diagnosis (PGD). It is eugenics without the terrible baggage that the word has carried from past diabolical experiments (think Mengele and the Nazis). But that does not mean that the method has not been misused. PGD is being used surreptitiously to select for sex particularly in India and China even though selecting for gender is banned there. It is estimated that as many as 10 million females have “disappeared” from PGD, abortion, infanticide, or neglect of female children3.
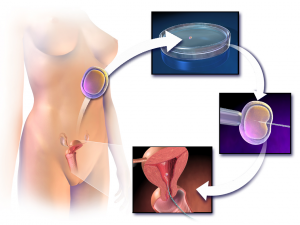 Diagram of in vitro fertilization – Wikipedia
Diagram of in vitro fertilization – Wikipedia
According to Mukherjee, there have been three principles that guide doctors in deciding which embryos will not be implanted during IVF. First, the gene needs to lead to a serious life-threatening disease with almost 100 percent chance of the child or adult developing the disease. Cystic fibrosis is a good example – a single gene causes the genetic disease. The disorder affects the lungs primarily, causing chronic coughing from frequent lung infections. Life expectancy is about 46. The misery is not limited to the lungs. Sinus infections, poor growth, clubbing of digits, fatty stools, and infertility (among males) are just some of the side effects. Second, the development of the gene will lead to “extraordinary suffering”. And finally, there must be a consensus among the medical community that the intervention is morally and ethically sound and the family involved has complete freedom of choice.
Even so, the Roman Catholic Church (and other religious institutions) has strongly objected to IVF and related gene technologies. John Hass, a Catholic theologian, states: “One reproductive technology which the Church has clearly and unequivocally judged to be immoral is in vitro fertilization or IVF. Unfortunately, most Catholics are not aware of the Church’s teaching, do not know that IVF is immoral, and some have used it in attempting to have children… In IVF, children are engendered through a technical process, subjected to “quality control,” and eliminated if found “defective.”4. Honestly, I don’t understand where this moral imperative comes from. If there is a God, He/She must have understood that we would eventually discover how to cure genetic diseases. Apparently Hass and the Church find no fault with technologies that would correct the problem after the embryo is in the womb but chaff at the idea of choosing to avoid the disease before the embryo is placed in the womb. I suspect that Hass might change his mind if he had to watch someone die slowly from a disease like cystic fibrosis5. Clearly, our society will continue to grapple with the ethical and moral issues of gene technologies particularly now that research is making social engineering theoretically “available”. Mukherjee discusses the identification of a gene related to psychic stress to emphasize how blurred the ethical decisions are potentially becoming. Where society draws the line is going to be as important as the genetic technology itself. But these ethical dilemmas are just the tip of the iceberg.
Improved safety and more careful oversight has gradually led to better research. New viruses have been developed to effectively deliver gene-altered DNA or RNA to cells that avoid catastrophic immune responses similar to what happened to Jesse Gelsinger. In 2014, viral delivery systems successfully treated hemophilia – the genetic disorder that prevents blood from clotting. And although the setback that genetic engineering suffered in the 1990s in the aftermath of Jesse’s death had been overcome as the new millennium approached, germ-line therapy was set back again when George W. Bush drastically reduced the use of ES cells in federal research programs in 2001. Germ-line therapy is the modification of the human genome in reproductive cells so that the modified gene is passed on to offspring. Imagine ridding genomes of gene mutations that cause cystic fibrosis or breast cancer (BRCA1) forever in families. Yet because ES cells are frequently obtained from embryos left over from IVF, Bush clamped down on the research (presumably based on pressure from the religious right) which nearly extinguished United States progress in the field for nearly a decade. I understand the abortion debate, but collecting ES cells from embryos that will never be implanted in woman’s womb seems to be carrying the abortion issue to drastic extremes.
Jennifer Doudna of the University of California, Berkeley and Emmanuelle Charpentier of the Helmholtz Centre for Infection Research knew from earlier research that bacteria had RNA that could find and recognize DNA in a virus and then deliver a protein which cut the virus DNA, thus disabling it – an effective way bacteria fought off viral attacks. By 2012, they were not only able to program the process to seek and cut any specified section of DNA, but they learned how to flood the region near the cut with desired DNA fragments that the cut DNA incorporated into its genome. In effect, they had created a gene splicing technique they designated CRISPR/Cas96 (clustered regularly interspaced short palindromic repeats). In other words, Doudna and Charpentier had discovered a means to exchange a serious mutant gene like the cystic fibrosis gene with a harmless gene. The dawn of genetic editing had begun7.
About the same time that Doudna and Charpentier were developing the CRISPR technology, scientists at Cambridge, England and at the Israeli Weizmann Institute were discovering how to make ES cells into primordial germ cells – these are the cells that develop into the sperm and egg in the embryo. The brave new world predicted by Huxley nearly 100 years ago in 1932 is upon us. The technology is now available to form a germ line cell which can be genetically modified with CRISPR technology. The modified cells can then be converted to sperm and eggs to form an embryo which will produce a genetically modified human through IVF – a transgenic human. However, as you might imagine, there are strict controls and bans on this research in the United States based on ethical and moral issues. Scientists are forbidden to introduce genetically modified cells that will develop into embryos directly into humans and ES cells cannot be genetically modified if they will form into sperm and egg cells. Most other countries have followed the US lead with similar bans. Mukherjee tries to explain the concern: “The crux, then, is not genetic emancipation (freedom from the bounds of hereditary illness), but genetic enhancement (freedom from the current boundaries of form and fate encoded by the human genome). The distinction between the two is the fragile pivot on which the future of genome editing whirls.” It is clear that we are wrestling with our past history of the misplaced promotion of horrible eugenics programs. I asked Doudna to clarify the reason for a moratorium: “the moratorium is not a call to outright ban engineering of the human germ line. Instead, it suggests a halt to such clinical use until a broader cross section of scientific, clinical, ethical, and regulatory experts, as well as the public at large have a chance to fully consider the ramifications.”
But we may not have the luxury of waiting until the ethics and morals of the science are thoroughly debated. In 2015, Junjiu Huang and his team at Sun Yat-sen University in Guangzhou, China, used CRISPR to eliminate a gene that causes a blood disorder in human embryos. There were problems in the products and the procedure was stopped (although there was never any intention of allowing the embryos to mature in a womb). The experiments set off international alarms and the scientific journals Nature, Cell, and Science refused to publish the paper. The paper was eventually published in Protein + Cell. Huang has made it clear that he will continue to pursue experiments to correct problems that surfaced during the previous work. “They did the research ethically” noted Tetsuya Ishii of Hokkaido University in Sapporo, Japan in Science, but several genetic watchdog groups called for an end to the procedures. Other scientists including a Nobel laureate were not disturbed by the research as long as the experiments were limited to clinical applications8.
 Genetic editing in human embryos.
Genetic editing in human embryos.
The incident with Junjiu Huang reminds me of the work that has been done on game theory. As far back as the 1920s one of the leading lights in mathematics, John von Neumann at the Institute of Advanced Study where Albert Einstein and Kurt Gödel worked (closely associated with Princeton University), sought to define, through mathematical expressions, logical procedures in games that could be applied to real-life scenarios. In his superb book Prisoner’s Dilemma: John von Neuman, Game theory, and the Puzzle of the Bomb, William Poundstone summarizes von Neumann’s work: “Von Neumann demonstrated mathematically that there is always a rational course of action for games of two players, provided their interests are completely opposed9.” One of the early applications of work on game theory came when the United States was deciding to build a hydrogen bomb – a huge leap in destructive capabilities compared to the atomic bomb. Many prominent scientists, such as Robert Oppenheimer, the director of the Manhattan Project, were outspoken against it. Seemingly they reasoned, the best strategy would be to cooperate with the Soviet Union whereby both countries would agree not to develop the H-bomb. The research was expensive and it would generate thousands of bombs that would be stockpiled and probably never be used. Game theory logic did not concur. There was only one possible step according to the logic of “game” brinkmanship between the US and the Soviets – build the H-bomb no matter whether the Soviets were willing to agree to a moratorium or not. There was simply no way to be absolutely sure the Soviets would live up to any potential agreement.
I think the same strategy is true with germ-line experiments. The logic is clear – it seems the Chinese are going to develop the technology regardless of what we do and not having the technology while other countries do could be detrimental to the best interests of the United States. The value of developing germ-line therapy seems even more crucial than, say, the H-bomb because the therapy will potentially lead to cures for horrible genetic diseases. I recognize the need to be discreet and careful, but we also need not dally on something so important. In December of 2015, the International Summit on Human Gene Editing was sponsored by the US National Academy of Sciences, the US Academy of Medicine, the Chinese Academy of Sciences, and the Royal Society of London. The planning committee summarized recommendations for “the development and human applications of genome editing” with agreements made to have future summits. The recommendations can be reviewed in an editorial by Theodore Friedmann in Molecular Therapy10. All I can say is that the sides are talking, and that is important. The research continues with some controls.
- In Jesse’s case, the gene was not inherited but was caused by a mutation in only one cell before birth. The result was unusual in that not all of his cells were OTC deficient as might be expected if he had inherited the trait. ↩
- Mukherjee, S. (2016) The Gene: An Intimate History, Scribner ↩
- see ref. 2 ↩
- see for example, Haas, J. M. (1998) Begotten not made: A catholic view of reproductive technology ↩
- I was raised a Roman Catholic, and I know that Catholics believe in divine inspiration. That is, they believe the Pope with or without the input of his advisers makes a decision on the morality of the issue with the understanding that the decision is inspired directly by God. I would hasten to point out that the terrorists that took down the World Trade Center believed they were divinely inspired also so believing does not make it so. I sometimes wonder if these men (and I emphasize men because there are no women in the upper echelons of the Holy See) ever wonder if their opinions are really divinely inspired. They place a great deal of confidence in a decision that will bring immense misery into the world – consider all those Catholics that refuse to use IVF and have children with serious genetic disorders ↩
- The Cas9 was the protein that performed the cutting. ↩
- see Exterminating invasive species with gene drives ↩
- Kaiser, J. and Normile, D. (2015) Embryo engineering study splits scientific community: Science, 348, 486-487 ↩
- Poundstone, W. (1992) Prisoner’s Dilemma: John von Neuman, Game theory, and the Puzzle of the Bomb: Anchor Books ↩
- Friedmann, T. (2016) An ASGCT Perspective on the National Academies Genome Editing Summit: Molecular Therapy, 24, 1-2 ↩
